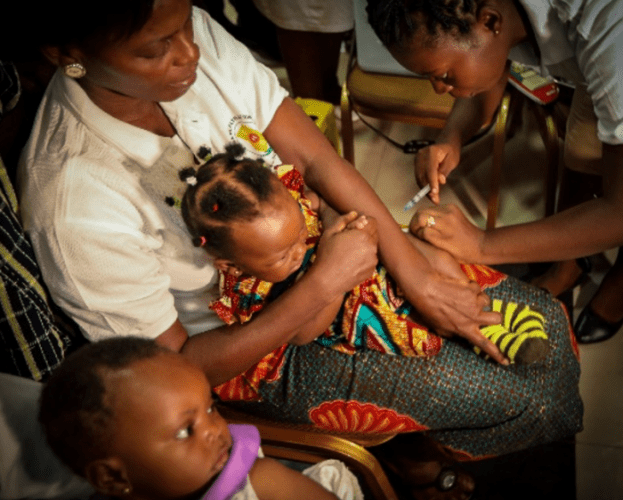Thirty years in the making, RTS,S, the malaria vaccine, also known by its brand name, Mosquirix, targets Plasmodium falciparum, the most common and most lethal of four malaria parasite species. It is an answer to a dire need. After decades of declining numbers of cases and deaths, the fight against malaria has stalled. Parasites resistant to the most widely used treatment, called artemisinin-based combination therapy, are spreading, while malaria mosquitoes are increasingly resistant to insecticides. And yet the rollout in Malawi and in two other African countries, isn’t quite the breakthrough the field has been waiting for. Mosquirix’s efficacy and durability are mediocre: Four doses offer only 30% protection against severe malaria, for no more than 4 years. Some experts question whether that is worth the cost and effort.
The biggest concerns, however, are about the vaccine’s safety. In the largest trial, children who received Mosquirix had a risk of meningitis 10 times higher than those who received a control vaccine. Mosquirix may not have triggered the meningitis cases—there are other possible explanations—but the possible risk worried the global health community so much that, rather than rolling out the vaccine across Africa, the World Health Organization (WHO) has decided to set up a pilot in Malawi, Ghana, and Kenya in which the vaccine will be given to hundreds of thousands of children.
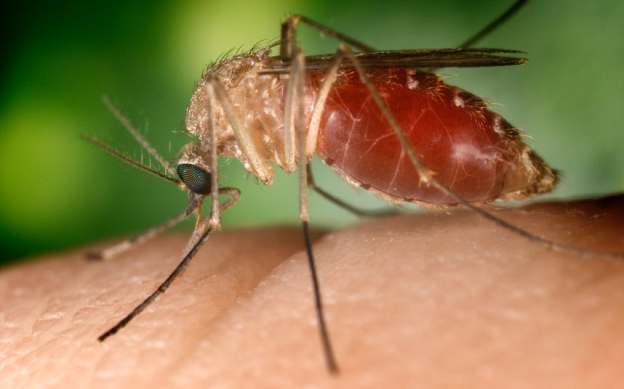
The Malaria parasite is a challenging target for a vaccine. It has a complex life cycle that begins when an infected female mosquito bites a human and spits Plasmodium cells called sporozoites into the bloodstream. They multiply in the liver, emerge as another cell type named merozoites, invade red blood cells, and continue to multiply. The blood cells burst, causing fever, headache, chills, muscle aches, and often anemia. (They also flood the blood with gametocytes—the parasite’s reproductive cells—ready to be picked up by the next mosquito.) Along the way, the parasite frequently changes its surface proteins. That makes it an elusive target for the immune system, and for a vaccine.
Mosquirix, developed in the 1980s by a team in Belgium at SmithKline-RIT, now part of GlaxoSmithKline (GSK), stimulates an immune response against a protein that occurs only on the sporozoites’ surface. To bolster the response, the research team fused the vaccine protein with a hepatitis B surface protein and added an adjuvant….
A relative outside, Danish anthropologist and vaccine researcher Peter Aaby of the Bandim Health Project in Guinea-Bissau, offered another argument against introduction. After reanalyzing the data from the biggest trial, Aaby discovered that although the vaccinated children had malaria less often, they did not die less often. Among girls, overall mortality was almost doubled, Aaby told his colleagues at the meeting. “This vaccine is killing girls,” he recalls saying. Whereas WHO expects the vaccine to save one life per 200 children vaccinated, Aaby believes one in 200 will die as a result of it; he predicts “a nightmare.”
Aaby and Christine Stabell Benn, a global health professor at the University of Southern Denmark, have an explanation. The married couple has studied routine vaccinations in Africa for decades and believes vaccines can “train” the immune system in ways that don’t affect just the target disease. Vaccines that contain a living, weakened pathogen—such as the vaccines against measles and tuberculosis—strengthen the immune system generally, Aaby and Stabell Benn say, making recipients better able to fight off other infections. But vaccines that contain a killed pathogen or only bits of it weaken the immune system, their theory goes—especially in girls, because their immune systems seem to respond more strongly to vaccines in general.
Excerpts from Jop de Vrieze, A Shot of Hope, Science Magazine, Nov. 29, 2019, at 1063