A plan to release over 750 million genetically modified mosquitoes into the Florida Keys in 2021 and 2022 received final approval from local authorities, against the objection of many local residents and a coalition of environmental advocacy groups. The proposal had already won state and federal approval.
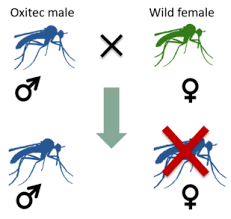
Approved by the Environment Protection Agency in May 2020, the pilot project is designed to test if a genetically modified mosquito is a viable alternative to spraying insecticides to control the Aedes aegypti. It’s a species of mosquito that carries several deadly diseases, such as Zika, dengue, chikungunya and yellow fever. The mosquito, named OX5034, has been altered to produce female offspring that die in the larval stage, well before hatching and growing large enough to bite and spread disease. Only the female mosquito bites for blood, which she needs to mature her eggs. Males feed only on nectar, and are thus not a carrier for disease. The mosquito also won federal approval to be released into Harris County, Texas, beginning in 2021, according to Oxitec, the US-owned, British-based company that developed the genetically modified organism (GMO)…
In 2009 and 2010, local outbreaks of dengue feverleft the Florida Keys Mosquito Control District desperate for new options. Despite an avalanche of effort — from aerial, truck and backpack spraying to the use of mosquito-eating fish — local control efforts to contain the Aedes aegypti with larvicide and pesticide had been largely ineffective.
And costly, too. Even though Aedes aegypti is only 1% of its mosquito population, Florida Keys Mosquito Control typically budgets more than $1 million a year, a full tenth of its total funding, to fighting it…
The new male mosquito, OX5034, is programmed to kill only female mosquitoes, with males surviving for multiple generations and passing along the modified genes to subsequent male offspring….Environmental groups worry that the spread of the genetically modified male genes into the wild population could potentially harm threatened and endangered species of birds, insects and mammals that feed on the mosquitoes.
Excerpt from Sandee LaMotte, 750 million genetically engineered mosquitoes approved for release in Florida Keys, CNN,