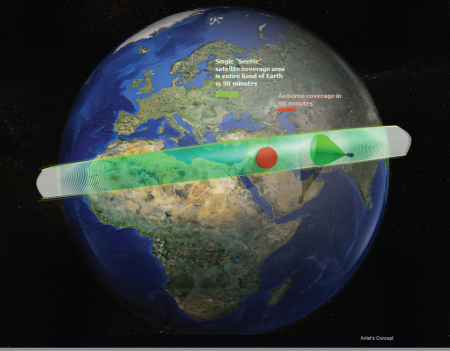
DARPA has selected seven teams of researchers to begin work on the Agency’s Electrical Prescriptions (ElectRx) program, which has as its goal the development of a closed-loop system that treats diseases by modulating the activity of peripheral nerves…. Ultimately, the program envisions a complete system that can be tested in human clinical trials aimed at conditions such as chronic pain, inflammatory disease, post-traumatic stress and other illnesses that may not be responsive to traditional treatments.
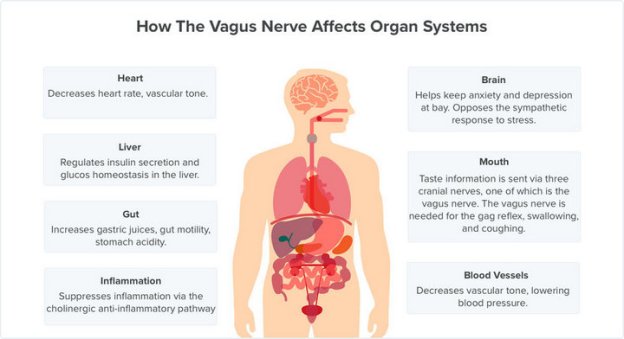
“The peripheral nervous system is the body’s information superhighway, communicating a vast array of sensory and motor signals that monitor our health status and effect changes in brain and organ functions to keep us healthy,“ said Doug Weber, the ElectRx program manager and a biomedical engineer who previously worked as a researcher for the Department of Veterans Affairs. “We envision technology that can detect the onset of disease and react automatically to restore health by stimulating peripheral nerves to modulate functions in the brain, spinal cord and internal organs.”
The oldest and simplest example of this concept is the cardiac pacemaker, which uses brief pulses of electricity to stimulate the heart to beat at a healthy rate. Extending this concept to other organs like the spleen may offer new opportunities for treating inflammatory diseases such as rheumatoid arthritis. Fighting inflammation may also provide new treatments for depression, which growing evidence suggests might be caused in part by excess levels of inflammatory biomolecules. Peripheral nerve stimulation may also be used to regulate production of neurochemicals that regulate learning and memory in the brain, offering new treatments for post-traumatic stress and other mental health disorders.
Circuit Therapeutics (Menlo Park, Calif.), a start-up co-founded by Karl Deisseroth and Scott Delp, is a new DARPA performer. The team plans to further develop its experimental optogenetic methods for treating neuropathic pain, building toward testing in animal models before seeking to move to clinical trials in humans.
A team at Columbia University (New York), led by Elisa Konofagou, will pursue fundamental science to support the use of non-invasive, targeted ultrasound for neuromodulation. The team aims to elucidate the underlying mechanisms that may make ultrasound an option for chronic intervention, including activation and inhibition of nerves.
A team at the Florey Institute of Neuroscience and Mental Health (Parkville, Australia), led by John Furness, is a first-time DARPA performer. Team members will seek to map the nerve pathways that underlie intestinal inflammation, with a focus on determining the correlations between animal models and human neural circuitry. They will also explore the use of neurostimulation technologies based on the cochlear implant —developed by Cochlear, Inc. to treat hearing loss, but adapted to modulate activity of the vagus nerve in response to biofeedback signals—as a possible treatment for inflammatory bowel disease.
A team at the Johns Hopkins University (Baltimore), led by Jiande Chen, aims to explore the root mechanisms of inflammatory bowel disease and the impact of sacral nerve stimulation on its progression. The team will apply a first-of-its-kind approach to visualize intestinal responses to neuromodulation in animal models.
A team at the Massachusetts Institute of Technology (Cambridge, Mass.), led by Polina Anikeeva, will aim to advance its established work in magnetic nanoparticles for localized, precision in vivo neuromodulation through thermal activation of neurons in animal models. The team’s work will target the adrenal gland and the splanchnic nerve circuits that govern its function. To increase specificity and minimize potential side effects of this method of stimulation, the team seeks to develop nanoparticles with the ability to bind to neuronal membranes. Dr. Anikeeva was previously a DARPA Young Faculty Awardee.
A team at Purdue University (West Lafayette, Ind.), led by Pedro Irazoqui, will leverage an existing collaboration with Cyberonics to study inflammation of the gastrointestinal tract and its responsiveness to vagal nerve stimulation through the neck. Validation of the mechanistic insights that emerge from the effort will take place in pre-clinical models in which novel neuromodulation devices will be applied to reduce inflammation in a feedback-controlled manner. Later stages of the effort could advance the design of clinical neuromodulation devices.
A team at the University of Texas, Dallas, led by Robert Rennaker and Michael Kilgard, will examine the use of vagal nerve stimulation to induce neural plasticity for the treatment of post-traumatic stress. As envisioned, stimulation could enhance learned behavioral responses that reduce fear and anxiety when presented with traumatic cues. Dr. Rennaker is a U.S. Marine Corps veteran who served in Liberia, Kuwait and Yugoslavia.
“Using the peripheral nervous system as a medium for delivering therapy is largely new territory and it’s rich with potential to manage many of the conditions that impact the readiness of our military and, more generally, the health of the nation,” Weber said. “It will be an exciting path forward.”
Press Release: Work Begins to Support Self-Healing of Body and Mind
Integrated, international efforts under ElectRx program blend mapping of neural circuits and development of novel bio-electrical interfaces OUTREACH@DARPA.MIL, Oct. 5, 2015









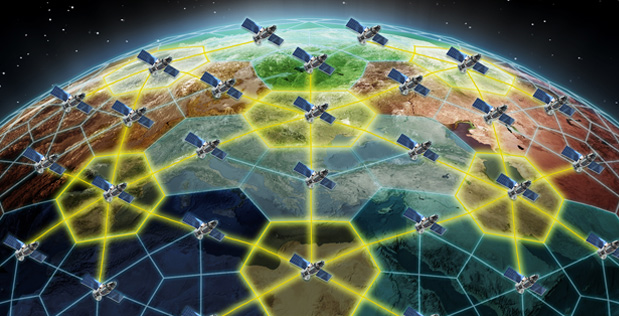












 The activity, hostile action that falls short of–but often precedes–violence, is sometimes referred to as gray zone warfare, the ‘zone’ being a sort of liminal state in between peace and war. The actors that work in it are difficult to identify and their aims hard to predict, by design…
The activity, hostile action that falls short of–but often precedes–violence, is sometimes referred to as gray zone warfare, the ‘zone’ being a sort of liminal state in between peace and war. The actors that work in it are difficult to identify and their aims hard to predict, by design…
 The world’s vast oceans and seas offer seemingly endless spaces in which adversaries of the United States can maneuver undetected. The U.S. military deploys networks of manned and unmanned platforms and sensors to monitor adversary activity, but the scale of the task is daunting and hardware alone cannot meet every need in the dynamic marine environment. Sea life, however, offers a potential new advantage. Marine organisms are highly attuned to their surroundings—their survival depends on it—and a new program out of DARPA’s Biological Technologies Office aims to tap into [marine animals] natural sensing capabilities to detect and signal when activities of interest occur in strategic waters such as straits and littoral regions.
The world’s vast oceans and seas offer seemingly endless spaces in which adversaries of the United States can maneuver undetected. The U.S. military deploys networks of manned and unmanned platforms and sensors to monitor adversary activity, but the scale of the task is daunting and hardware alone cannot meet every need in the dynamic marine environment. Sea life, however, offers a potential new advantage. Marine organisms are highly attuned to their surroundings—their survival depends on it—and a new program out of DARPA’s Biological Technologies Office aims to tap into [marine animals] natural sensing capabilities to detect and signal when activities of interest occur in strategic waters such as straits and littoral regions.

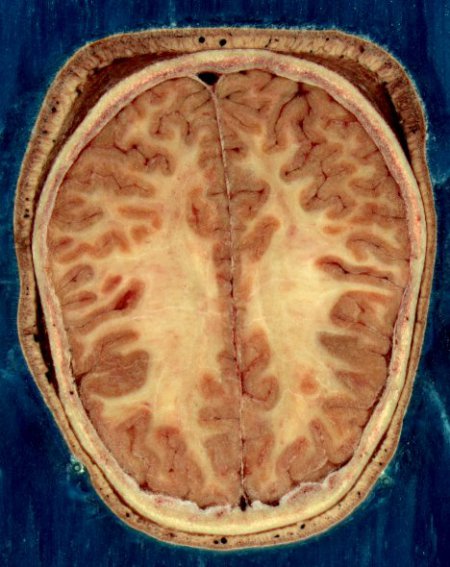
 Two millivolts is enough to show that someone has seen something even before he knows he has seen it himself. The two millivolts in question are those associated with P300, a fleeting electrical signal produced by a human brain which has just recognised an object it has been seeking. Crucially, this signal is detectable by electrodes in contact with a person’s scalp before he is consciously aware of having recognised anything.
Two millivolts is enough to show that someone has seen something even before he knows he has seen it himself. The two millivolts in question are those associated with P300, a fleeting electrical signal produced by a human brain which has just recognised an object it has been seeking. Crucially, this signal is detectable by electrodes in contact with a person’s scalp before he is consciously aware of having recognised anything.